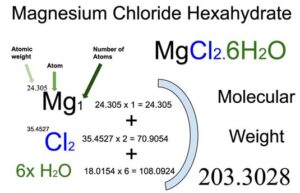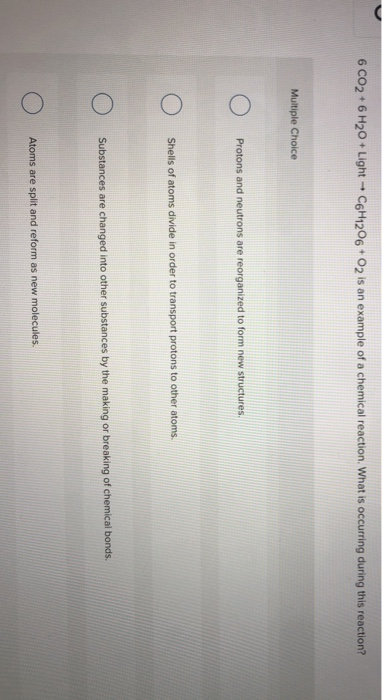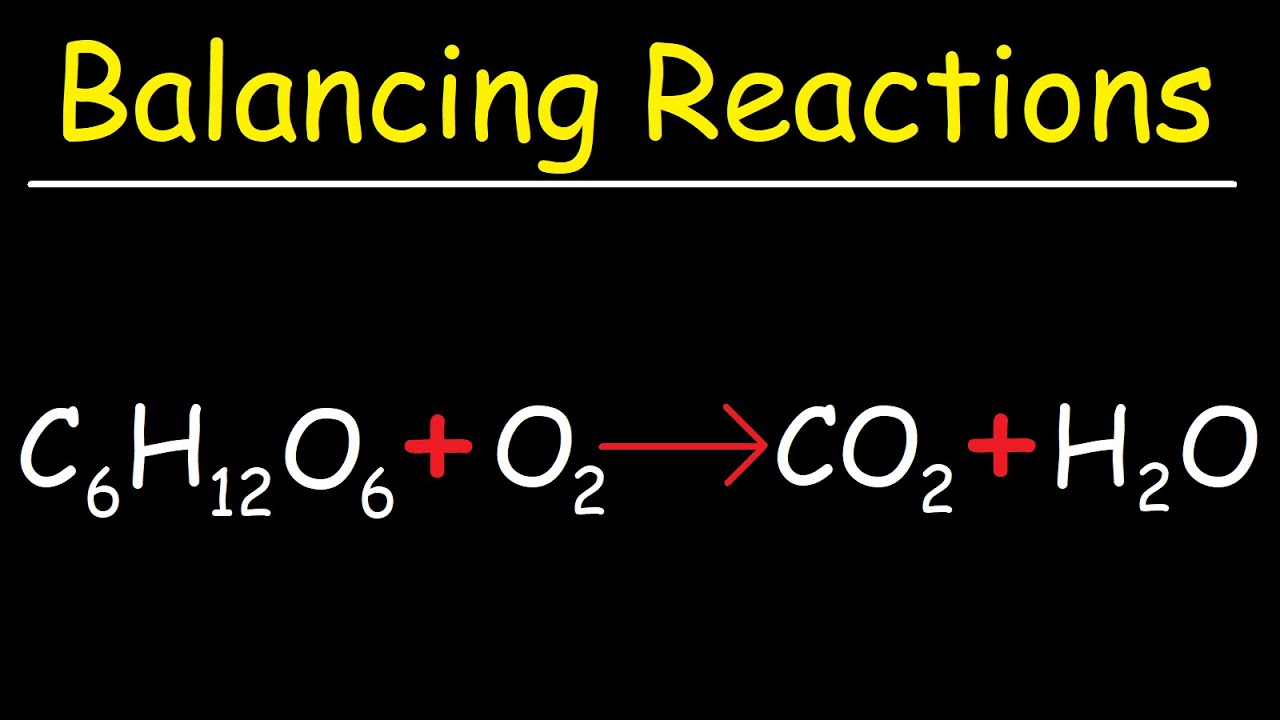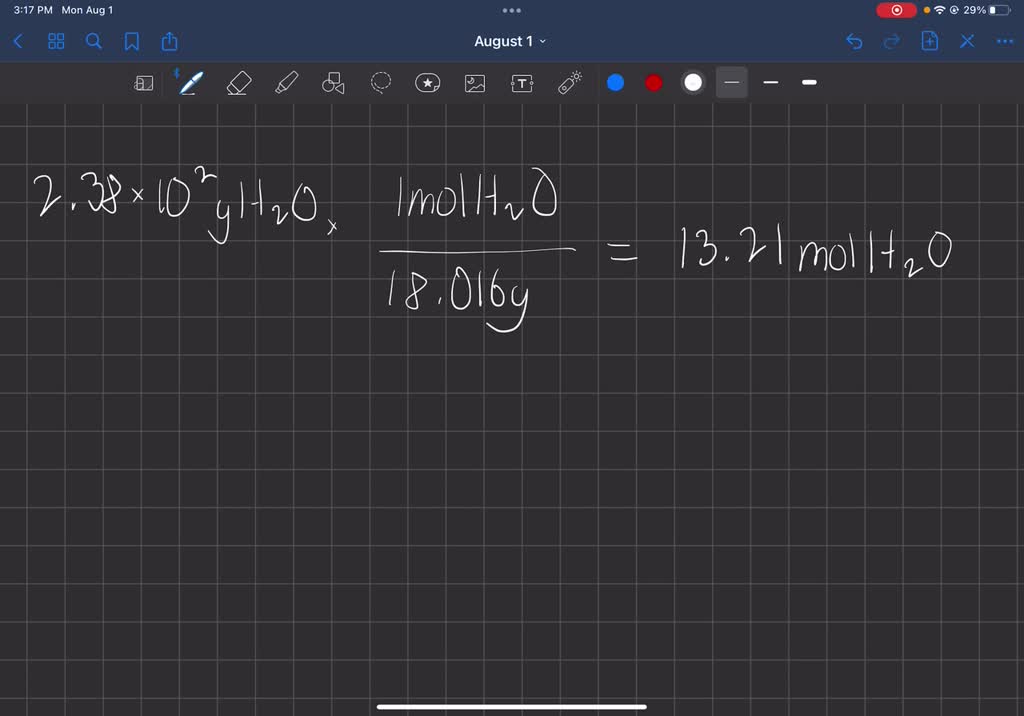
SOLVED: Consider this reaction: 6 CO2 + 6 H2O + light C6H12O6 + 6 O2If there were 2.38 x 102 g of H2O, 18.6 moles of CO2, and plenty of light, what would be the theoretical yield of the reaction?
عبدالرحمن العتيبي on Twitter: "A simple example of a combustion reaction is the burning of methane: CH4(g) + 2 O2(g) → CO2(g) + 2 H2O(g)" / Twitter

The structure diagrams of (H2O)6⁻ cluster isomers. The red and white... | Download Scientific Diagram
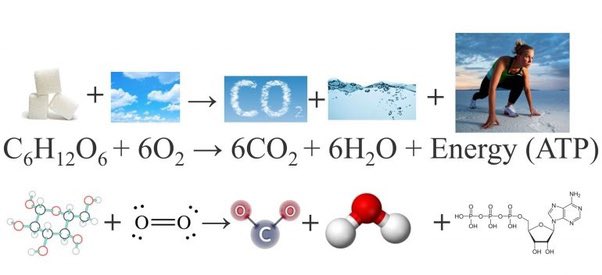
If C6H12O6 + 6O2 + 6H2O — >6CO2 + 12H2O + energy is a balanced equation then what is the formula of energy when everything gets balanced ?

Optimized Cluster Structures of trans-Carboxyl (1Ht) with 6 to 11 H2O... | Download Scientific Diagram

Consider this reaction: 6 CO2 + 6 H2O + light equation C6H12O6 + 6 O2 If there were 2.38 x 102 g of - Brainly.in
![SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [ SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [](https://cdn.numerade.com/ask_previews/e7bf0f40-8942-4c62-a0e8-90258b6ac48b_large.jpg)
SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [

For the reaction: Co(H2O)6^{2+} + 4Cl^- \leftrightharpoons CoCl4^{2-} + 6H2O After adding 12M HCl, what would be the roles of H^+ ions and Cl^- ions? | Homework.Study.com
![Cation Exchange Reactions in Layered Double Hydroxides Intercalated with Sulfate and Alkaline Cations (A(H2O)6)[M2+6Al3(OH)18(SO4)2]·6H2O (M2+ = Mn, Mg, Zn; A+ = Li, Na, K) | Journal of the American Chemical Society Cation Exchange Reactions in Layered Double Hydroxides Intercalated with Sulfate and Alkaline Cations (A(H2O)6)[M2+6Al3(OH)18(SO4)2]·6H2O (M2+ = Mn, Mg, Zn; A+ = Li, Na, K) | Journal of the American Chemical Society](https://pubs.acs.org/cms/10.1021/jacs.8b11389/asset/images/jacs.8b11389.social.jpeg_v03)