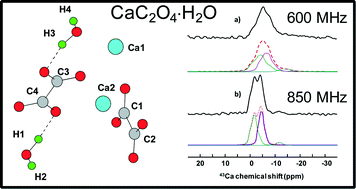
1 Chapter 3 The principle of TGA and its applications: TGA is thermogravimetric analysis. It is one of the thermal method of an
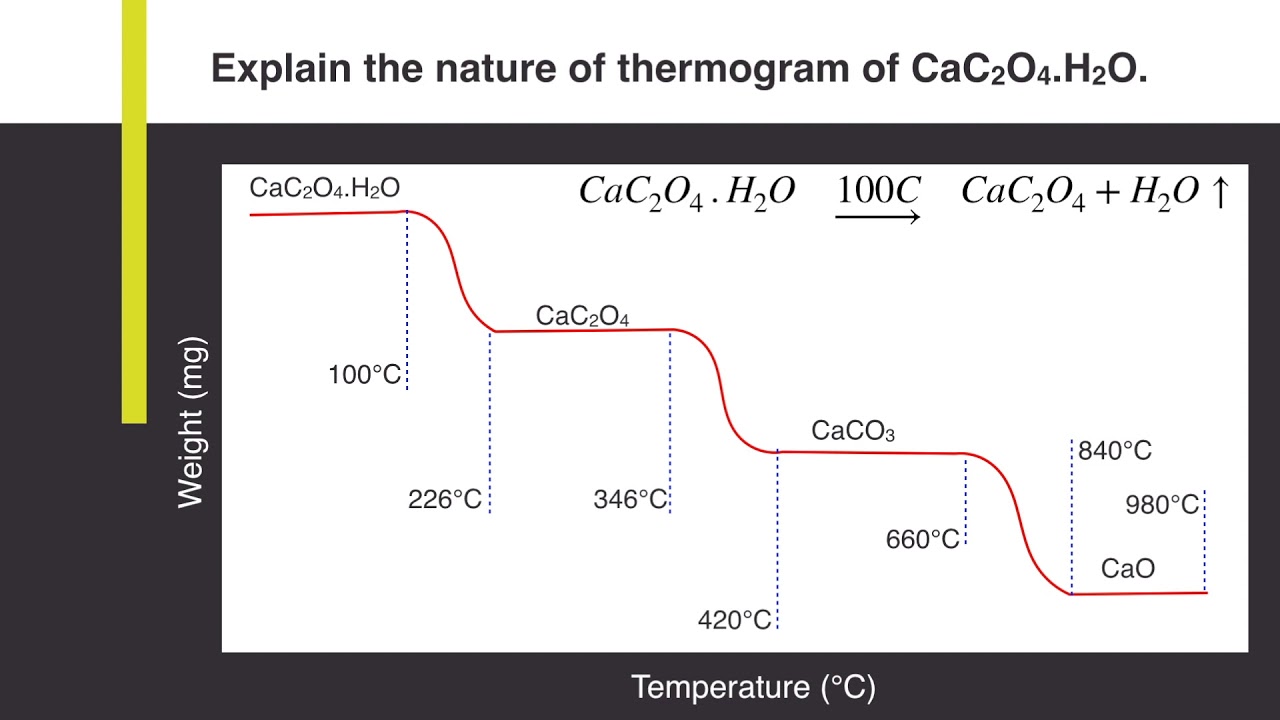
Explain the nature of thermogram of Calcium Oxalate Monohydrate (CaC2O4.H2O) | Analytical Chemistry - YouTube

Computational prediction of the pattern of thermal gravimetry data for the thermal decomposition of calcium oxalate monohydrate – topic of research paper in Chemical sciences. Download scholarly article PDF and read for

Thermal Decomposition of CaC2O4·H2O Studied by Thermo-Raman Spectroscopy with TGA/DTA | Analytical Chemistry
Thermal Stability of Calcium Oxalates from CO2 Sequestration for Storage Purposes: An In-Situ HT-XRPD and TGA Combined Study

Solubility of Calcium Oxalate Monohydrate in Concentrated Electrolyte Solutions | Journal of Chemical & Engineering Data
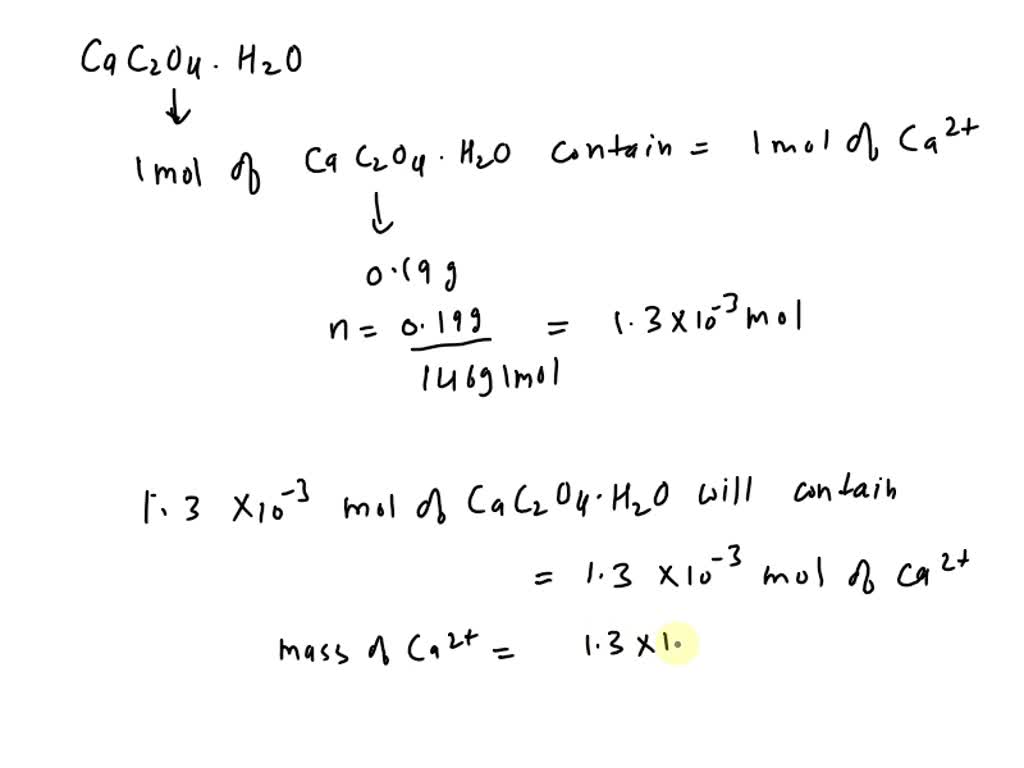
SOLVED: What is the concentration of calcium (Ca2+) in calcium oxalate monohydrate (CaC2O4.H2O). Ca2+(aq) + C2O42-(aq) + H2O (l) → CaC2O4·H2O (s) The mass of calcium oxalate monohydrate is 0.190grams. what is